Cell-Free DNA Technology for NIPT
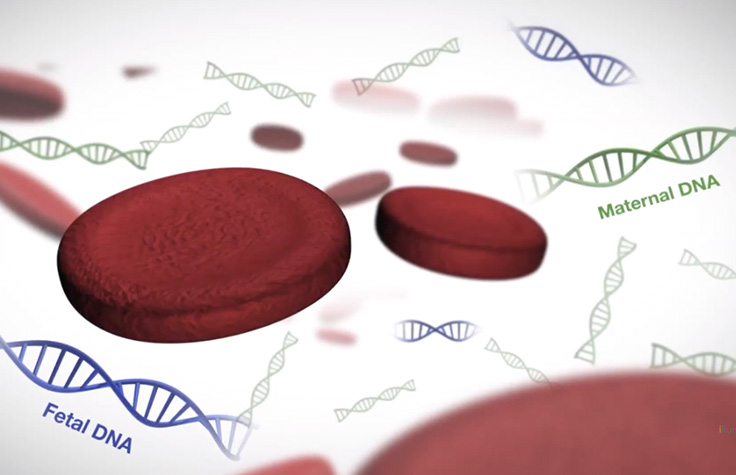
What is Cell-Free DNA?
Cell-free DNA are short fragments of DNA released into the bloodstream through a natural process of cell death. During pregnancy, the mother’s blood contains cell-free DNA (cfDNA), both from her own tissue, and from the fetus via the placenta. Approximately 2-20% of total cfDNA in maternal blood is placental.1,2 cfDNA derived from the placenta can be detected after 7+ weeks gestation and is undetectable within hours postpartum.2
A noninvasive prenatal test (NIPT) analyzes cfDNA from a maternal blood sample to screen for common chromosomal conditions in the fetus.

Fetal Fraction
The percentage of total cell-free DNA (cfDNA) in a sample derived from the fetus is called the fetal fraction—which can affect the ability of NIPT to detect fetal aneuploidy. When considering various cfDNA NIPT technologies, it’s important to understand how fetal fraction is used and how it can affect test results.
Fetal Fraction and Test Failure
cfDNA NIPT technologies based on microarrays and targeted sequencing employ a minimum fetal fraction cutoff. If the fetal fraction of a sample is below this threshold, no result (a test failure) is reported. Strict fetal fraction cutoffs lead to higher test failure rates, even upon redraw.
Additionally, labs are often not reimbursed when a test does not return a result even though they have incurred the costs associated with running the test.
Dynamic Thresholds
Whole-genome sequencing-based technology can employ dynamic thresholds, which provide an alternative to strict fetal fraction cutoffs. This approach allows samples with low fetal fraction to be reported, but only if there is sufficient sequencing data to make a confident call. The intent is to minimize cfDNA NIPT failure rates while ensuring accurate calls. The Illumina VeriSeq NIPT Solution* offers a dynamic threshold testing metric called iFACT.
* For In Vitro Diagnostic Use. Contact an Illumina representative for regional availability.

Cell-free DNA Screening Beyond the Common Trisomies
Expanding cell-free DNA screening to more women and a broader range of conditions has transformed the prenatal screening environment, raising questions about who should be offered screening, for what, by whom, and how.
We're hosting a fireside chat with two users of cell-free DNA screening from diverse systems for a relaxed conversation about their own experiences.
View WebinarAdditional Resources
Interested in receiving newsletters, case studies, and information on new applications? Enter your email address.
References
- Barrett, A, Zimmerman BG, Wang D, Holloway A, Chitty L. Implementing prenatal diagnosis based on cell-free fetal DNA: Accurate identification of factors affecting fetal DNA yield. PLoS One. 2011;6(10):e25202.
- Nigam A, Saxena P, Prakash A, Acharya A.Detection of fetal nucleic acid in maternal plasma: A novel noninvasive prenatal diagnostic technique. J Interntl Med Sci Acad. 2012; 25(3): 119-120.