High-Throughput Sequencing

Bringing greater insights, answers, and breakthroughs to light
See what is possible through the latest advances in Illumina high-throughput sequencing technology
Advantages of High-Throughput Sequencing
The latest innovations in Illumina high-throughput sequencing technology power advanced sequencing applications to generate dramatically more data for more impactful insights. Increasingly, scientists are leveraging the advantages of advanced next-generation sequencing (NGS) applications with our simplified, cost-efficient high-throughput sequencing workflows.

Improve Statistical Power
Increased statistical power from larger sample sizes for more impactful conclusions.
Access Previously Missed Information
See the full breadth of variation with whole-genome or whole-transcriptome sequencing.
Get Multidimensional Insights
Multidimensional insights with sequencing provide a single readout from multiple -omes across DNA, RNA, and protein.
Increase Analytical Resolution
Transition to high-resolution approaches, including single-cell sequencing and spatial analyses for insights into complex tissues.
High-ThrougHput Sequencing Technology Innovations
The latest advances in Illumina sequencing have enabled high-intensity sequencing applications, bringing insights that were unimaginable even a few years ago and removing barriers with every technological innovation.
Scalable
Get an unparalleled breadth of targets. Create multi-dimensional insights with optimized indexing strategies. Illumina high-throughput sequencing provides a single robust readout for multiple -omes.
Cost-Effective
Amplify your insights. Cast a wider net with larger sample sizes. Cost-effective high-throughput sequencing opens up your options to do high-intensity sequencing applications and gain additional insights for a more comprehensive view of your sample.
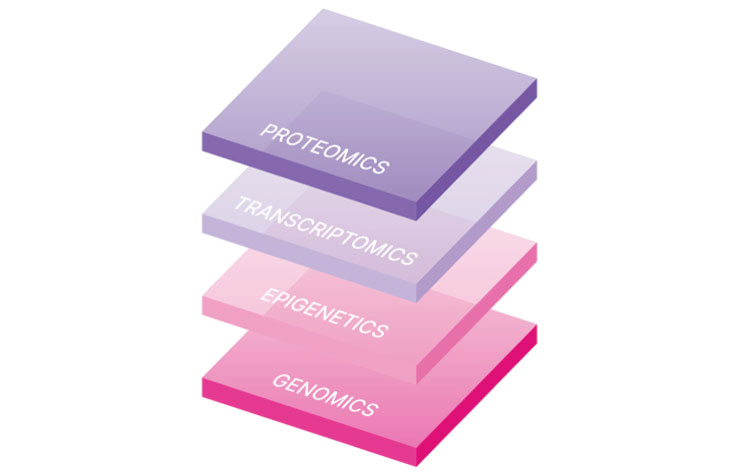
Discover More with Multiomics
Combine genomic data with data from other modalities such as transcriptomics, epigenetics, and proteomics to measure gene expression, gene activation, and protein levels. High-throughput omics profiling studies enable a more comprehensive understanding of molecular changes contributing to normal development, cellular response, and disease.
Popular High-Throughput Sequencing Methods
Achieve impactful insights and accelerate your lab’s work with these methods.
Explore common high-throughput NGS methods and their key benefits.
Genomics
Whole-Genome Sequencing
This method delivers a high-resolution view of the entire genome, ideal for discovery applications.
Long-Read Sequencing
Long reads can help scientists resolve challenging regions of the genome, detect complex structural variants, and perform de novo assembly.
Whole-Exome Sequencing
This approach involves sequencing the protein-coding regions of the genome to uncover genetic influences on disease and population health.
Epigenetics
Methylation Sequencing
Methylation sequencing approaches provide insight into cytosine methylation patterns at a single nucleotide level.
ATAC-Seq
The assay for transposase-accessible chromatin with sequencing (ATAC-Seq) is a rapid, sensitive method for profiling accessible chromatin across the genome.
Transcriptomics
CITE-Seq
Cellular indexing of transcriptomes and epitopes (CITE-Seq) simultaneously quantifies gene expression and surface protein expression levels at single-cell resolution.
Total RNA-Seq
This method involves sequencing both coding RNA and multiple forms of noncoding RNA for a comprehensive view of the transcriptome.
High-Throughput Sequencers
Dream big. The NovaSeq X Series can make it happen.
Unleash your ambitions with the NovaSeq X and NovaSeq X Plus Sequencing Systems, our most powerful and most sustainable sequencers yet. The ultra-high-throughput NovaSeq X Plus System delivers up to 16 Tb output and 52B single reads per dual flow cell run. Advanced chemistry, optics, and informatics provide exceptional accuracy and speed that enable transformative economics for groundbreaking discoveries.
View System View ReagentsHigh-Throughput Sequencers

Drive discoveries with the NovaSeq 6000 system
A tried-and-true high-throughput instrument with improved economics and an output of up to 6 Tb and 20B single reads in < 2 days. Unlock a full spectrum of genetic variation and biological function with high-throughput sequencing on the NovaSeq 6000 system. Advances in reagents support a vast breadth of targets and complex indexing strategies.
View System View Reagents
Accessibility for new and emerging applications with the NextSeq 2000 system
NextSeq 2000 System is redesigned from the ground up to maximize future proofing, offering sequencing power for high-throughput applications. With adjustible output and scalability for evolving needs and larger studies, the system and reagents make a vast variety of data-rich multiomics applications accessible, including whole-exome, and single-cell sequencing.

High-Throughput NGS Data Analysis
Perform ultra-rapid, user-friendly data analysis
Illumina DRAGEN Secondary Analysis
The platform for ultra-rapid secondary genomic analysis with highly accurate results, diverse applications, and frequent updates. DRAGEN can process NGS data for an entire human genome at 30x coverage in about 25 minutes.
Learn More About DRAGENAggregate, explore, and share large volumes of data
Illumina Connected Analytics
This comprehensive cloud-based data management and analysis software platform empowers researchers to manage, analyze, and interpret large volumes of multiomics data in a secure, scalable, and flexible environment. The platform is built for high-volume operations limited by data silos and a lack of bioinformatics and data science tools.
Learn More About Illumina Connected AnalyticsHigh-throughput lab setup
Streamline Your Lab Operations
The NovaSeq X Series provides a new standard in operational simplicity, driving breakthroughs and maximizing productivity.
Bioinformatics Pipeline Setup
Find out how to simplify the informatics infrastructure and data analysis pipeline setup process and access more data for more insights.
Multiplex Sequencing
Pool and sequence large numbers of NGS libraries simultaneously during a single run.
Supporting Genome Center Growth
The Deakin Genomics Centre uses Illumina NGS for high-throughput sequencing projects to lower sequencing costs and provide a faster sequencing data turnaround time.
Identifying Breast Cancer Targets
Garvan Institute scientists use Illumina high-throughput NGS to sequence more than a million breast cancer cells to help create a comprehensive cellular atlas of breast cancer.
Automate Your Library Prep
These walk-away automation methods for Illumina kits eliminate the need to develop your own method and can help you reduce hands-on time and minimize errors.
Featured Webinars

Deploy High-Throughput Proteomics with NGS
Learn how to take multiplex proteomics to the next level and discover protein biomarkers using Illumina sequencers.

Omics-Based Precision Medicine Methods
Whole-genome sequencing is an increasingly viable approach for precision diagnosis in clinical care.
Related Resources
Trade In Your HiSeq System
Interested in trading in your HiSeq System for the latest high-throughput sequencer with more discovery power? You may be eligible to trade in your existing instrument for a new NovaSeq 6000 System.
Everything You Need at a Glance
Explore Illumina high-throughput sequencing options. Get additional training, find software, plan experiments, purchase, and get support.